Which State Of Matter Is The Only One That Has A Fixed Shape?
3.3: Classifying Affair According to Its State—Solid, Liquid, and Gas
- Folio ID
- 47454
- To describe the solid, liquid and gas phases.
Water can take many forms. At low temperatures (below \(0^\text{o} \text{C}\)), it is a solid. When at "normal" temperatures (between \(0^\text{o} \text{C}\) and \(100^\text{o} \text{C}\)), it is a liquid. While at temperatures above \(100^\text{o} \text{C}\), water is a gas (steam). The state that water is in depends upon the temperature. Each land has its own unique gear up of physical backdrop. Matter typically exists in one of 3 states: solid, liquid, or gas.



The state that a given substance exhibits is also a concrete belongings. Some substances exist every bit gases at room temperature (oxygen and carbon dioxide), while others, like h2o and mercury metal, exist as liquids. Most metals exist as solids at room temperature. All substances tin can exist in whatever of these three states. Figure \(\PageIndex{2}\) shows the differences amidst solids, liquids, and gases at the molecular level. A solid has definite volume and shape, a liquid has a definite book merely no definite shape, and a gas has neither a definite volume nor shape.
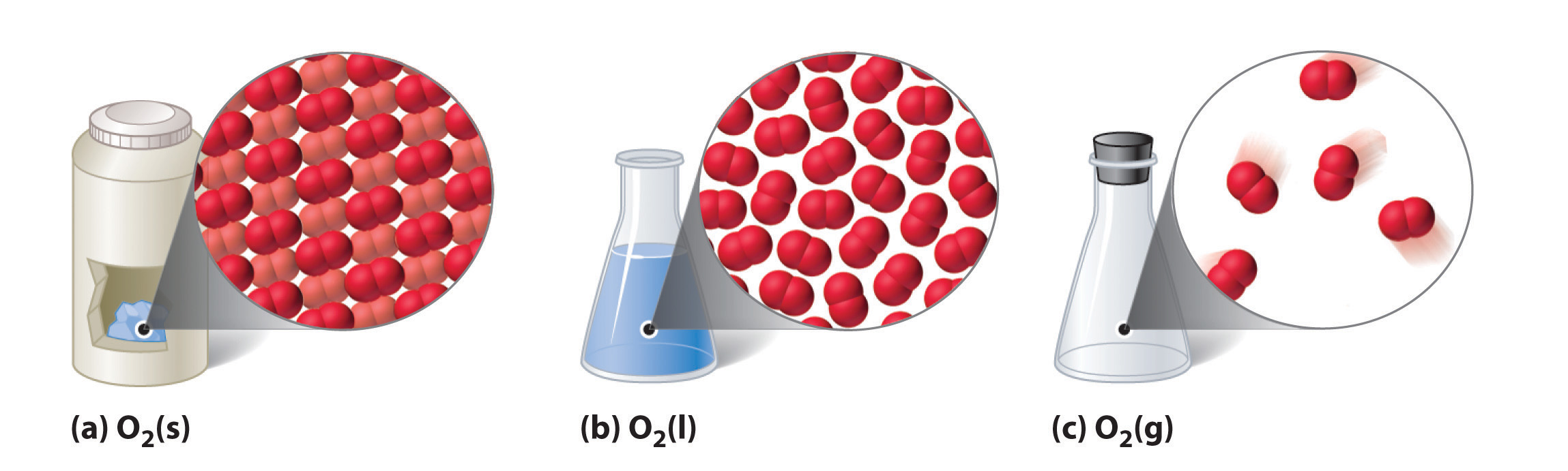
Technically speaking, a fourth country of affair chosen plasma exists, but it does not naturally occur on earth, so we will omit it from our study hither.
.jpg?revision=1&size=bestfit&width=197&height=196)
A plasma globe operating in a darkened room. (CC By-SA 3.0; Chocolateoak).
Solids
In the solid state, the individual particles of a substance are in fixed positions with respect to each other considering at that place is not enough thermal energy to overcome the intermolecular interactions between the particles. Every bit a consequence, solids have a definite shape and volume. Most solids are difficult, only some (like waxes) are relatively soft. Many solids equanimous of ions can too exist quite brittle.
Solids are defined by the following characteristics:
- Definite shape (rigid)
- Definite volume
- Particles vibrate effectually fixed axes
If we were to cool liquid mercury to its freezing betoken of \(-39^\text{o} \text{C}\), and under the right pressure conditions, we would notice all of the liquid particles would go into the solid country. Mercury tin can be solidified when its temperature is brought to its freezing point. Nevertheless, when returned to room temperature conditions, mercury does not exist in solid state for long, and returns back to its more mutual liquid form.
Solids usually have their constituent particles arranged in a regular, three-dimensional array of alternating positive and negative ions called a crystal. The consequence of this regular arrangement of particles is sometimes visible macroscopically, equally shown in Figure \(\PageIndex{three}\). Some solids, especially those composed of large molecules, cannot hands organize their particles in such regular crystals and be as amorphous (literally, "without form") solids. Glass is one instance of an amorphous solid.
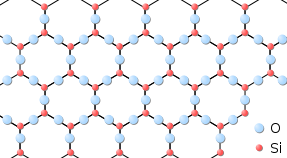
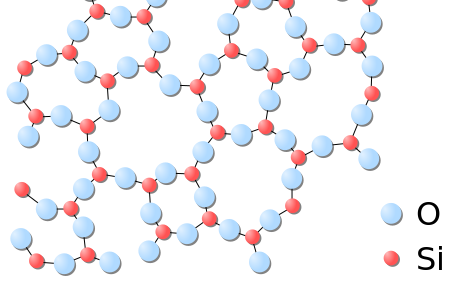
Liquids
If the particles of a substance have plenty energy to partially overcome intermolecular interactions, so the particles can move virtually each other while remaining in contact. This describes the liquid state. In a liquid, the particles are however in close contact, and so liquids have a definite volume. However, because the particles tin move about each other rather freely, a liquid has no definite shape and takes a shape dictated by its container.
Liquids have the following characteristics:
- No definite shape (takes the shape of its container).
- Has definite volume.
- Particles are gratuitous to move over each other, but are still attracted to each other.
A familiar liquid is mercury metal. Mercury is an bibelot. It is the only metal we know of that is liquid at room temperature. Mercury too has an power to stick to itself (surface tension)—a property that all liquids exhibit. Mercury has a relatively high surface tension, which makes it very unique. Here you run across mercury in its common liquid form.
Video \(\PageIndex{1}\): Mercury humid to become a gas.
If we rut liquid mercury to its boiling signal of \(357^\text{o} \text{C}\) under the right pressure conditions, we would find all particles in the liquid state get into the gas state.
Gases
If the particles of a substance have enough energy to completely overcome intermolecular interactions, and then the particles can separate from each other and move near randomly in infinite. This describes the gas state, which we will consider in more detail elsewhere. Like liquids, gases have no definite shape, merely different solids and liquids, gases take no definite volume either. The alter from solid to liquid commonly does not significantly change the volume of a substance. However, the change from a liquid to a gas significantly increases the volume of a substance, by a factor of ane,000 or more. Gases have the following characteristics:
- No definite shape (takes the shape of its container)
- No definite volume
- Particles movement in random motion with little or no attraction to each other
- Highly compressible
| Characteristics | Solids | Liquids | Gases |
|---|---|---|---|
| shape | definite | indefinite | indefinite |
| volume | definite | definite | indefinite |
| relative intermolecular interaction strength | strong | moderate | weak |
| relative particle positions | in contact and stock-still in place | in contact simply not fixed | non in contact, random positions |
What state or states of affair does each argument, describe?
- This country has a definite volume, but no definite shape.
- This state has no definite volume.
- This state allows the individual particles to move most while remaining in contact.
Solution
- This statement describes the liquid state.
- This argument describes the gas state.
- This argument describes the liquid state.
What land or states of matter does each statement describe?
- This state has individual particles in a fixed position with regard to each other.
- This country has individual particles far apart from each other in space.
- This land has a definite shape.
- Respond a:
- solid
- Answer b:
- gas
- Answer c:
- solid
Summary
- Iii states of matter exist—solid, liquid, and gas.
- Solids accept a definite shape and volume.
- Liquids accept a definite volume, but take the shape of the container.
- Gases have no definite shape or volume.
Which State Of Matter Is The Only One That Has A Fixed Shape?,
Source: https://chem.libretexts.org/Bookshelves/Introductory_Chemistry/Map%3A_Introductory_Chemistry_(Tro)/03%3A_Matter_and_Energy/3.03%3A_Classifying_Matter_According_to_Its_StateSolid_Liquid_and_Gas
Posted by: lunaupellift.blogspot.com


0 Response to "Which State Of Matter Is The Only One That Has A Fixed Shape?"
Post a Comment