Can Metals Form Covalent Bonds
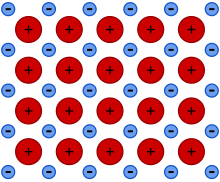
An case showing metallic bonding. + represents cations, - represents the free floating electrons.
Metallic bonding is a type of chemical bonding that arises from the electrostatic attractive forcefulness between conduction electrons (in the grade of an electron cloud of delocalized electrons) and positively charged metal ions. It may be described as the sharing of gratis electrons amid a structure of positively charged ions (cations). Metallic bonding accounts for many physical properties of metals, such as strength, ductility, thermal and electrical resistivity and conductivity, opacity, and luster.[ane] [2] [3] [4]
Metallic bonding is non the but type of chemical bonding a metal tin can exhibit, even as a pure substance. For example, elemental gallium consists of covalently-jump pairs of atoms in both liquid and solid-land—these pairs grade a crystal structure with metallic bonding between them. Another example of a metal–metal covalent bond is the mercurous ion (Hg 2+
2 ).
History
Every bit chemistry developed into a science, it became clear that metals formed the majority of the periodic tabular array of the elements, and great progress was made in the description of the salts that can be formed in reactions with acids. With the appearance of electrochemistry, it became clear that metals generally go into solution as positively charged ions, and the oxidation reactions of the metals became well understood in their electrochemical series. A picture emerged of metals equally positive ions held together by an ocean of negative electrons.
With the appearance of quantum mechanics, this moving-picture show was given a more formal interpretation in the form of the free electron model and its further extension, the nearly free electron model. In both models, the electrons are seen as a gas traveling through the construction of the solid with an free energy that is substantially isotropic, in that information technology depends on the foursquare of the magnitude, not the direction of the momentum vector k. In three-dimensional k-space, the gear up of points of the highest filled levels (the Fermi surface) should therefore be a sphere. In the most-free model, box-like Brillouin zones are added to thousand-space by the periodic potential experienced from the (ionic) structure, thus mildly breaking the isotropy.
The advent of Ten-ray diffraction and thermal analysis made it possible to study the structure of crystalline solids, including metals and their alloys; and phase diagrams were developed. Despite all this progress, the nature of intermetallic compounds and alloys largely remained a mystery and their written report was often only empirical. Chemists generally steered abroad from anything that did not seem to follow Dalton'south laws of multiple proportions; and the trouble was considered the domain of a different science, metallurgy.
The well-nigh-free electron model was eagerly taken up by some researchers in this field, notably Hume-Rothery, in an try to explicate why certain intermetallic alloys with certain compositions would form and others would non. Initially Hume-Rothery's attempts were quite successful. His idea was to add together electrons to inflate the spherical Fermi-balloon inside the series of Brillouin-boxes and determine when a certain box would exist full. This predicted a fairly large number of alloy compositions that were afterwards observed. Every bit soon every bit cyclotron resonance became available and the shape of the balloon could be adamant, it was constitute that the assumption that the airship was spherical did not hold, except possibly in the case of caesium. This finding reduced many of the conclusions to examples of how a model can sometimes give a whole series of correct predictions, yet still be wrong.
The nigh-gratuitous electron debacle showed researchers that whatever model that causeless that ions were in a sea of costless electrons needed modification. So, a number of breakthrough mechanical models—such as band structure calculations based on molecular orbitals or the density functional theory—were adult. In these models, one either departs from the atomic orbitals of neutral atoms that share their electrons or (in the case of density functional theory) departs from the total electron density. The free-electron motion-picture show has, all the same, remained a dominant one in pedagogy.
The electronic band structure model became a major focus not only for the study of metals merely fifty-fifty more so for the study of semiconductors. Together with the electronic states, the vibrational states were likewise shown to form bands. Rudolf Peierls showed that, in the instance of a one-dimensional row of metal atoms—say, hydrogen—an instability had to arise that would lead to the breakup of such a chain into individual molecules. This sparked an interest in the general question: when is collective metal bonding stable and when will a more localized form of bonding take its place? Much research went into the report of clustering of metal atoms.
As powerful every bit the concept of the band structure model proved to be in describing metal bonding, it has the drawback of remaining a i-electron approximation of a many-torso trouble. In other words, the energy states of each electron are described every bit if all the other electrons but form a homogeneous groundwork. Researchers such as Mott and Hubbard realized that this was peradventure appropriate for strongly delocalized s- and p-electrons; but for d-electrons, and even more for f-electrons, the interaction with electrons (and atomic displacements) in the local environment may become stronger than the delocalization that leads to broad bands. Thus, the transition from localized unpaired electrons to itinerant ones partaking in metallic bonding became more comprehensible.
The nature of metallic bonding
The combination of two phenomena gives rise to metal bonding: delocalization of electrons and the availability of a far larger number of delocalized energy states than of delocalized electrons.[ description needed ] The latter could be called electron deficiency.
In 2nd
Graphene is an instance of 2-dimensional metallic bonding. Its metallic bonds are like to effluvious bonding in benzene, naphthalene, anthracene, ovalene, etc.
In 3D
Metallic aromaticity in metallic clusters is some other example of delocalization, this fourth dimension often in three-dimensional arrangements. Metals take the delocalization principle to its extreme, and one could say that a crystal of a metallic represents a single molecule over which all conduction electrons are delocalized in all three dimensions. This means that inside the metallic ane can generally not distinguish molecules, and then that the metallic bonding is neither intra- nor inter-molecular. 'Nonmolecular' would perhaps be a improve term. Metallic bonding is generally not-polar, because even in alloys there is trivial difference amid the electronegativities of the atoms participating in the bonding interaction (and, in pure elemental metals, none at all). Thus, metallic bonding is an extremely delocalized communal grade of covalent bonding. In a sense, metallic bonding is not a 'new' blazon of bonding at all. Information technology describes the bonding merely equally present in a clamper of condensed matter: be it crystalline solid, liquid, or even glass. Metallic vapors, in contrast, are oft diminutive (Hg) or at times incorporate molecules, such as Na2, held together by a more than conventional covalent bond. This is why it is not correct to speak of a single 'metallic bail'.[ description needed ]
Delocalization is nearly pronounced for s- and p-electrons. Delocalization in caesium is so strong that the electrons are virtually freed from the caesium atoms to form a gas constrained but past the surface of the metal. For caesium, therefore, the picture of Cs+ ions held together by a negatively charged electron gas is not inaccurate.[a] For other elements the electrons are less free, in that they still experience the potential of the metal atoms, sometimes quite strongly. They crave a more intricate breakthrough mechanical handling (east.g., tight binding) in which the atoms are viewed as neutral, much like the carbon atoms in benzene. For d- and peculiarly f-electrons the delocalization is not strong at all and this explains why these electrons are able to continue behaving as unpaired electrons that retain their spin, adding interesting magnetic properties to these metals.
Electron deficiency and mobility
Metal atoms contain few electrons in their valence shells relative to their periods or energy levels. They are electron-scarce elements and the communal sharing does not change that. At that place remain far more bachelor free energy states than there are shared electrons. Both requirements for conductivity are therefore fulfilled: strong delocalization and partly filled energy bands. Such electrons can therefore easily alter from one energy state to a slightly different one. Thus, not merely do they become delocalized, forming a sea of electrons permeating the construction, merely they are also able to migrate through the construction when an external electrical field is applied, leading to electrical conductivity. Without the field, at that place are electrons moving every bit in all directions. Inside such a field, some electrons will adjust their state slightly, adopting a different moving ridge vector. Consequently, there will be more moving one way than some other and a cyberspace current will consequence.
The freedom of electrons to migrate also gives metallic atoms, or layers of them, the capacity to slide past each other. Locally, bonds can easily be broken and replaced by new ones after a deformation. This process does non affect the communal metallic bonding very much, which gives ascent to metals' characteristic malleability and ductility. This is specially truthful for pure elements. In the presence of dissolved impurities, the normally easily formed cleavages may be blocked and the material go harder. Aureate, for instance, is very soft in pure form (24-karat), which is why alloys are preferred in jewelry.
Metals are typically also good conductors of heat, simply the conduction electrons only contribute partly to this phenomenon. Collective (i.eastward., delocalized) vibrations of the atoms, known as phonons that travel through the solid as a wave, are bigger contributors.
However, a substance such as diamond, which conducts estrus quite well, is non an electrical conductor. This is not a result of delocalization being absent in diamond, but merely that carbon is not electron deficient.
Electron deficiency is important in distinguishing metallic from more conventional covalent bonding. Thus, nosotros should amend the expression given above to: Metallic bonding is an extremely delocalized communal form of electron-scarce[b] covalent bonding.
Metal radius
The metallic radius is defined as one-half of the distance betwixt the ii adjacent metal ions in the metallic structure. This radius depends on the nature of the atom likewise as its environment—specifically, on the coordination number (CN), which in turn depends on the temperature and practical pressure.
When comparing periodic trends in the size of atoms it is frequently desirable to apply the and so-called Goldschmidt correction, which converts diminutive radii to the values the atoms would accept if they were 12-coordinated. Since metallic radii are largest for the highest coordination number, correction for less dense coordinations involves multiplying by 10, where 0 < x < 1. Specifically, for CN = 4, x = 0.88; for CN = vi, x = 0.96, and for CN = viii, x = 0.97. The correction is named after Victor Goldschmidt who obtained the numerical values quoted to a higher place.[6]
The radii follow general periodic trends: they decrease across the period due to the increment in the effective nuclear charge, which is not offset by the increased number of valence electrons; but the radii increase down the group due to an increment in the main quantum number. Between the 4d and 5d elements, the lanthanide contraction is observed—there is very little increment of the radius down the group due to the presence of poorly shielding f orbitals.
Strength of the bail
The atoms in metals take a strong attractive force between them. Much energy is required to overcome it. Therefore, metals oftentimes have high boiling points, with tungsten (5828 Yard) being extremely loftier. A remarkable exception is the elements of the zinc group: Zn, Cd, and Hg. Their electron configurations end in ...ns 2, which resembles a noble gas configuration, similar that of helium, more than and more than when going down the periodic table, considering the energy differential to the empty due northp orbitals becomes larger. These metals are therefore relatively volatile, and are avoided in ultra-high vacuum systems.
Otherwise, metallic bonding can be very strong, even in molten metals, such as gallium. Even though gallium will melt from the heat of i's hand simply above room temperature, its boiling indicate is not far from that of copper. Molten gallium is, therefore, a very nonvolatile liquid, thanks to its strong metallic bonding.
The potent bonding of metals in liquid grade demonstrates that the energy of a metallic bond is not highly dependent on the direction of the bond; this lack of bond directionality is a directly upshot of electron delocalization, and is all-time understood in contrast to the directional bonding of covalent bonds. The energy of a metallic bail is thus mostly a function of the number of electrons which surround the metal cantlet, as exemplified by the embedded cantlet model.[vii] This typically results in metals assuming relatively simple, close-packed crystal structures, such as FCC, BCC, and HCP.
Given high enough cooling rates and appropriate alloy composition, metal bonding can occur even in spectacles, which have amorphous structures.
Much biochemistry is mediated past the weak interaction of metal ions and biomolecules. Such interactions, and their associated conformational changes, have been measured using dual polarisation interferometry.
Solubility and compound germination
Metals are insoluble in water or organic solvents, unless they undergo a reaction with them. Typically, this is an oxidation reaction that robs the metal atoms of their itinerant electrons, destroying the metallic bonding. Still metals are often readily soluble in each other while retaining the metallic character of their bonding. Gilt, for example, dissolves easily in mercury, even at room temperature. Even in solid metals, the solubility can be extensive. If the structures of the two metals are the same, there tin can fifty-fifty be consummate solid solubility, as in the case of electrum, an alloy of silver and gold. At times, all the same, two metals volition grade alloys with unlike structures than either of the two parents. One could call these materials metal compounds. But, because materials with metallic bonding are typically non molecular, Dalton's law of integral proportions is not valid; and oftentimes a range of stoichiometric ratios can exist accomplished. Information technology is better to abandon such concepts as 'pure substance' or 'solute' in such cases and speak of phases instead. The written report of such phases has traditionally been more the domain of metallurgy than of chemical science, although the ii fields overlap considerably.
Localization and clustering: from bonding to bonds
The metal bonding in complex compounds does non necessarily involve all elective elements every bit. It is quite possible to have one or more elements that do non partake at all. Ane could flick the conduction electrons flowing around them like a river around an isle or a large stone. It is possible to find which elements do partake: east.g., by looking at the cadre levels in an X-ray photoelectron spectroscopy (XPS) spectrum. If an element partakes, its peaks tend to be skewed.
Some intermetallic materials, e.g., practice exhibit metallic clusters reminiscent of molecules; and these compounds are more a topic of chemistry than of metallurgy. The germination of the clusters could be seen as a fashion to 'condense out' (localize) the electron-scarce bonding into bonds of a more localized nature. Hydrogen is an farthermost example of this form of condensation. At loftier pressures it is a metal. The core of the planet Jupiter could exist said to be held together by a combination of metal bonding and high pressure induced by gravity. At lower pressures, however, the bonding becomes entirely localized into a regular covalent bail. The localization is and then complete that the (more than familiar) H2 gas results. A similar argument holds for an element such equally boron. Though it is electron-deficient compared to carbon, it does not form a metallic. Instead it has a number of circuitous structures in which icosahedral B12 clusters boss. Charge density waves are a related phenomenon.
As these phenomena involve the movement of the atoms toward or abroad from each other, they can be interpreted as the coupling betwixt the electronic and the vibrational states (i.e. the phonons) of the fabric. A different such electron-phonon interaction is thought to lead to a very dissimilar upshot at low temperatures, that of superconductivity. Rather than blocking the mobility of the charge carriers by forming electron pairs in localized bonds, Cooper-pairs are formed that no longer experience whatsoever resistance to their mobility.
Optical properties
The presence of an ocean of mobile charge carriers has profound effects on the optical properties of metals, which tin can just be understood by because the electrons as a collective, rather than because the states of individual electrons involved in more conventional covalent bonds.
Light consists of a combination of an electrical and a magnetic field. The electrical field is usually able to excite an elastic response from the electrons involved in the metallic bonding. The event is that photons cannot penetrate very far into the metal and are typically reflected, although some may also be captivated. This holds equally for all photons in the visible spectrum, which is why metals are often silverish white or grayish with the characteristic specular reflection of metallic luster. The balance between reflection and absorption determines how white or how grayness a metal is, although surface tarnish tin can obscure the luster. Argent, a metal with high electrical conductivity, is one of the whitest.
Notable exceptions are ruby copper and yellowish gold. The reason for their colour is that there is an upper limit to the frequency of the light that metallic electrons can readily respond to: the plasmon frequency. At the plasmon frequency, the frequency-dependent dielectric function of the free electron gas goes from negative (reflecting) to positive (transmitting); higher frequency photons are not reflected at the surface, and practice non contribute to the color of the metal. There are some materials, such as indium tin oxide (ITO), that are metallic conductors (really degenerate semiconductors) for which this threshold is in the infrared,[eight] which is why they are transparent in the visible, but good reflectors in the infrared.
For silver the limiting frequency is in the far ultraviolet, but for copper and gold it is closer to the visible. This explains the colors of these ii metals. At the surface of a metallic, resonance furnishings known as surface plasmons can outcome. They are collective oscillations of the conduction electrons, like a ripple in the electronic ocean. However, even if photons have enough energy, they usually practise non have enough momentum to ready the ripple in movement. Therefore, plasmons are hard to excite on a bulk metallic. This is why gold and copper wait like lustrous metals albeit with a dash of color. However, in colloidal gilded the metallic bonding is confined to a tiny metallic particle, which prevents the oscillation wave of the plasmon from 'running away'. The momentum selection rule is therefore broken, and the plasmon resonance causes an extremely intense absorption in the green, with a resulting purple-red colour. Such colors are orders of magnitude more intense than ordinary absorptions seen in dyes and the like, which involve private electrons and their energy states.
Run into also
- Atomic radii of the elements (data page)
- Bonding in solids – Nomenclature of bondings
- Metallic aromaticity – Concept of aromaticity extended to metals
Notes
- ^ If the electrons were truly free, their energy would only depend on the magnitude of their wave vector k, non its management. That is, in k-space, the Fermi level should form a perfect sphere. The shape of the Fermi level can exist measured by cyclotron resonance and is never a sphere, not fifty-fifty for caesium.[5]
- ^ Electron deficiency is a relative term: it ways fewer than half of the electrons needed to complete the next noble gas configuration. For instance, lithium is electron scarce with respect to neon, but electron-rich with respect to the previous element of group 0, helium.
References
- ^ Metallic bonding. chemguide.co.uk
- ^ Metal structures. chemguide.co.uk
- ^ Chemical Bonds. chemguide.co.uk
- ^ "Physics 133 Lecture Notes" Bound, 2004. Marion Campus. physics.ohio-state.edu
- ^ Okumura, K. & Templeton, I. M. (1965). "The Fermi Surface of Caesium". Proceedings of the Royal Society of London A. 287 (1408): 89–104. Bibcode:1965RSPSA.287...89O. doi:10.1098/rspa.1965.0170. JSTOR 2415064. S2CID 123127614.
- ^ Shriver and Atkins' Inorganic Chemical science. Oxford University Press. 2010. pp. 74–. ISBN978-0-nineteen-923617-6.
- ^ Daw, Murray Southward.; Foiles, Stephen M.; Baskes, Michael I. (1993). "The embedded-cantlet method: a review of theory and applications". Materials Science Reports (Submitted manuscript). ix (7–8): 251–310. doi:10.1016/0920-2307(93)90001-U.
- ^ Brewer, Scott H.; Franzen, Stefan (2002). "Indium Tin Oxide Plasma Frequency Dependence on Canvas Resistance and Surface Adlayers Determined past Reflectance FTIR Spectroscopy". The Periodical of Physical Chemistry B. 106 (50): 12986–12992. doi:10.1021/jp026600x.
Can Metals Form Covalent Bonds,
Source: https://en.wikipedia.org/wiki/Metallic_bonding
Posted by: lunaupellift.blogspot.com


0 Response to "Can Metals Form Covalent Bonds"
Post a Comment